Week 1: Introduction:
- History and Importance of phase diagram
Week 2: Thermodynamic formulation for single component phase diagram
- Free energy concept
- Free energy variation in single component system
Week 3: Phase diagram in single component system
- Single component phase diagram in water and CO2
Week 4: Solution thermodynamics
- Concept of idea solution and real solution
- Formulation of ideal solution and Chemical potential and equilibrium concept
Week 5: Solution thermodynamics and phase diagram in ideal binary solution
- Free energy composition diagram
- Evolution of isomorphous phase diagram
Week 6: Solution thermodynamics and quasichemical approach in binary solution
- Regular solution and quasichemical approach
- Free energy composition diagram in regular solution
Week 7: Phase diagrams in regular binary system
- Phase diagrams for different conditions of,
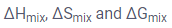
- Evolution of deep eutectic and congruent melting point
- Phase separation
Week 8: Examples of phase diagrams in binary system
- Eutectic
- Eutectoid
- Peritectic

DOWNLOAD APP
FOLLOW US